-for double award you only need to know the Haber Process, Single Award people, you also need to know the Contact Process for the manufacture of Sulphuric Acid!
5.21 recall that nitrogen from air, and hydrogen from natural gas or the cracking of hydrocarbons, are used in the manufacture of ammonia
So if they ask what are the raw materials used in the Haber process, you know it. It's nitrogen from the air and hydrogen either from natural gas, which is methane (CH4); or from the cracking of hydrocarbons. (Cracking is in the Crude Oil post.)
5.22 describe the manufacture of ammonia by the Haber process, including the essential conditions:
i. a temperature of about 450°C
ii. a pressure of about 200 atmospheres
iii. an iron catalyst
Remember these conditions!! And remember that the reaction is reversible. Also, the forwards reactions is exothermic.
N2 + 3H2 ⇌ 2NH3
So decreasing the temperature would actually increase the yield, however, it is still done at a fairly high temperature to speed up the reaction. It makes the rate of reaction faster so the manufacturers get their ammonia quicker, as they say, time is money. The reaction would be too slow otherwise at low temperatures. It would be useless to have a low temperature and achieve a high yield of ammonia if it's going to take ages. You need the gases to reach equilibrium within the very short time that they will be in contact with the catalyst in the reactor.So 450°C is a compromise, and still produces a reasonably high proportion of ammonia.
The catalyst does NOT affect the amount of products made. The yield of ammonia stays the same, you just get it faster because it speeds up the reaction by lowering the activation energy needed for the reaction. :)
The catalyst does NOT affect the amount of products made. The yield of ammonia stays the same, you just get it faster because it speeds up the reaction by lowering the activation energy needed for the reaction. :)
Increasing the pressure would favour the forwards reaction which is what is wanted, to get more ammonia. This is because if you look at the balanced equation, there are 4 moles of gas on the reactants side (left) but 2 moles of gas (ammonia) on the right hand side (products). So according to Le Chatelier's principle where you try to remove the change, if you increase pressure, the equlibrium would move to the right hand side to decrease pressure. And the products have less pressure because there are only 2 moles there.
Also, just as an extra, just thought that this will be useful to know and is very logical: :)
Also, just as an extra, just thought that this will be useful to know and is very logical: :)
Credit for the information goes to
http://www.chemguide.co.uk/physical/equilibria/haber.html
And no plagiarism was intended!
Rate considerations
Increasing the pressure brings the molecules closer together. In this particular instance, it will increase their chances of hitting and sticking to the surface of the catalyst where they can react. The higher the pressure the better in terms of the rate of a gas reaction.
Economic considerations
Very high pressures are very expensive to produce on two counts.
You have to build extremely strong pipes and containment vessels to withstand the very high pressure. That increases your capital costs when the plant is built.
High pressures cost a lot to produce and maintain. That means that the running costs of your plant are very high.
The compromise
200 atmospheres is a compromise pressure chosen on economic grounds. If the pressure used is too high, the cost of generating it exceeds the price you can get for the extra ammonia produced.
Economic considerations
Very high pressures are very expensive to produce on two counts.
You have to build extremely strong pipes and containment vessels to withstand the very high pressure. That increases your capital costs when the plant is built.
High pressures cost a lot to produce and maintain. That means that the running costs of your plant are very high.
The compromise
200 atmospheres is a compromise pressure chosen on economic grounds. If the pressure used is too high, the cost of generating it exceeds the price you can get for the extra ammonia produced.
5.23 understand how the cooling of the reaction mixture liquefies the ammonia produced and allows the unused hydrogen and nitrogen to be recirculated
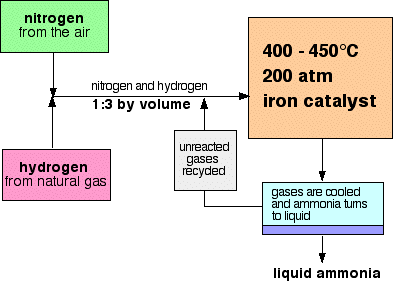
Separating the ammonia
When the gases leave the reactor they are hot and at a very high pressure. Ammonia is easily liquefied under pressure as long as it isn't too hot, and so the temperature of the mixture is lowered enough for the ammonia to turn to a liquid. The nitrogen and hydrogen remain as gases even under these high pressures, and can be recycled.
Recycling
At each pass of the gases through the reactor, only about 15% of the nitrogen and hydrogen converts to ammonia. (This figure also varies from plant to plant.) By continual recycling of the unreacted nitrogen and hydrogen, the overall conversion is about 98%.
5.24 recall the use of ammonia in the manufacture of nitric acid and fertilisers
So ammonia is used to make nitric acid and fertilisers, as you know from bio, plants need nitrates to grow.
Just in case you want to know, here are some properties of ammonia:
- alkaline gas (turns damp red litmus paper blue, which is the test for ammonia!)
- extremely soluble in water--it forms a weak alkali-->ammonia solution
- less dense than air
- colourless gas with pungent odour
HI MICHELLE :)
ReplyDeleteguess who
no
ReplyDeleteYou are awesome!
ReplyDeleteBy the way that she writes her comments 'anyways' 'in hopes' show that she maybe would not be very instructive on english igcse! ;). Thanks anyway, it is useful!
ReplyDeleteHahhaha okay well I'm just being casual. My English IGCSE scores were perfectly fine. And you're welcome, I'm glad!! :)
DeleteBefore you judge people, look at yourself first. I had a difficult time deciphering what you were saying.
DeleteYou made a mistake...
ReplyDeleteThe reaction is reversible!
ye, you're right
Deletethe arrow is meant to point in both directions like this :⇌
I did say the reaction was reversible, and the picture does have the arrow in both directions.. but I'll change the one I typed to the double arrow since you've kindly commented it. :)
DeleteIve got a goddy test tomorrow and i did not study and got pretty worried until i came across ur blog!!!!!! Thanks a zillion;-)
ReplyDeleteu cunts all suck
ReplyDeleteI completely agree
DeleteHey Michelle thanks for this, its really helpful , what school r u in
ReplyDeletethis was like a cup cake of knowledge, fast and easy.
ReplyDeleteThank you very much, in my book it was not explained too well :) thanks
ReplyDeleteThanks a lot :)
ReplyDeletethanks a bunch!! currently revising for the may/june 2021 series, this helped very much
ReplyDelete