For whoever asked for mole calculations. :)
It's detailed and has some example questions with working out, and some practice questions and tables for you to do too. Hope it helps!
Note that these are taken from my class book. They are not my materials.
It's detailed and has some example questions with working out, and some practice questions and tables for you to do too. Hope it helps!
Note that these are taken from my class book. They are not my materials.
1.15 calculate relative formula masses (Mr) from relative atomic masses (Ar)
Relative formula mass: the sum of masses of all the atoms in a formula
Relative atomic mass: the average mass of all the isotopes of an element
Examples:
Species
|
Formula
|
No. of atoms of each element in one molecule
|
Ar of each element
|
Sum of Ar
|
Relative Formula Mass (Mr)
|
Carbon dioxide
|
CO2
|
1 carbon
2 oxygen
|
C=12
O=16
|
12 + (16 x 2)
|
44
|
Oxygen
|
O2
|
2 oxygen
|
O=16
|
16 x 2
|
32
|
Aluminium oxide
|
Al2O3
|
2 aluminium
3 oxygen
|
Al=27
O=16
|
(27 x 2) + (16 x 3)
|
102
|
Aluminium sulphate
|
Al2(SO4)3
|
2 aluminium
3 sulphur
12 oxygen
|
Al=27
S=32
O=16
|
(27 x 2) + (32 x 3) + (16 x 12)
|
342
|
To calculate relative atomic mass (Ar): the sum of the relative abundance of each isotope multiplied by its mass number, divided by 100.
E.g. About 75% of all chlorine atoms have a mass number of 35 (18 neutrons), about 25% have a mass number of 37 (20 neutrons).
The relative atomic mass of chlorine is:
(75 x 35) + (25 x 37) / 100 = 35.5
1.18 carry out mole calculations using relative atomic mass (Ar) and relative formula mass (Mr)
n=m/Mr
Where n=number of moles, m=mass in grams and Mr=relative formula mass

e.g. how many moles are there in 120g of NaOH(s)?
n=m/Mr = 120g/40 = 3
There are 3 moles.
Try filling this out:
Substance
|
Formula
|
Mr
|
Mass of sample (g)
|
Number of moles in sample (n=m/Mr)
|
E.g. Water
|
H2O
|
18
|
9
|
0.5
|
Carbon dioxide
|
88
|
|||
Ammonia
|
1.7
|
|||
Sulphur dioxide
|
0.64
|
|||
Sulphur trioxide
|
80
|
|||
Hydrogen bromide
|
24.3
|
|||
Sulphuric acid
|
0.098
|
|||
Nitric acid
|
3.15
|
|||
Sodium nitrate
|
21.25
|
|||
Sodium carbonate
|
53
|
Example:
What mass of hydrogen is produced when 192g of magnesium is reacted with hydrochloric acid?
Step 1: write a balanced equation for the reaction:
Mg + 2HCl à MgCl2 + H2
Step 2: under the equation, fill in all the information you have been given, and also the Mr of the species involved:
Equation
|
Mg + 2HCl à MgCl2 + H2
|
|||
Mass
|
192g
|
This is the value you want to find
|
||
Mr
|
24
|
2
|
||
n
|
||||
Step 3: work out the number of moles of the reactant, using n=m/Mr then use the mole ratio of the equation to fill in the number of moles of the product:
Equation
|
Mg + 2HCl à MgCl2 + H2
|
|||
Mass
|
192g
|
This is the value you want to find
|
||
Mr
|
24
|
2
|
||
n
|
8
|
à
1:1 mole ratio
|
8
|
|
Step 4: now you have the Mr and number of moles of the product, rearrange n=m/Mr to find the mass:
m=n x Mr
Equation
|
Mg + 2HCl à MgCl2 + H2
|
|||
Mass
|
192g
|
16g
|
||
Mr
|
24
|
2
|
||
n
|
8
|
à
1:1 mole ratio
|
8
|
|
Once you are familiar with the process, you don't need a calculation frame, but keep your workings well ordered and logical.
- What mass of oxygen is needed to react with 8.5g of hydrogen sulphide (H2S)?
2H2S + 3O2 à 2SO2 + 2H2O
Find out how many moles of hydrogen sulphide there are, since you have its mass.
n=m/Mr
n=8.5/34=0.25
Mole ratio= 2:3 (2 moles of H2S : 3 moles of O2)
(0.25/2) x 3 = 0.375 --- 0.375 moles of O2 react with 0.25 moles of H2S, now you want to find the mass.
n=m/Mr
m= n x Mr
m= 0.375 x 32 = 12g
- Railway lines are welded together by the thermite reaction, which produces molten iron. What mass of iron is formed from 1kg of iron oxide?
Fe2O3 + 2Al à 2Fe + Al2O3
n=m/Mr
n=1000g/160=6.25
Mole ratio= 1:2
6.25 x 2=12.5
m=n x Mr
m= 12.5 x 56 = 700g
Practice:
- What mass of sodium hydroxide is formed when 46g of sodium reacts with excess water?
- Calculate the mass of water formed when 32g of oxygen reacts with excess hydrogen.
- Calcium carbonate thermally decomposes to form calcium oxide and carbon dioxide. If 44g of carbon dioxide are collected, what mass of calcium oxide is formed?
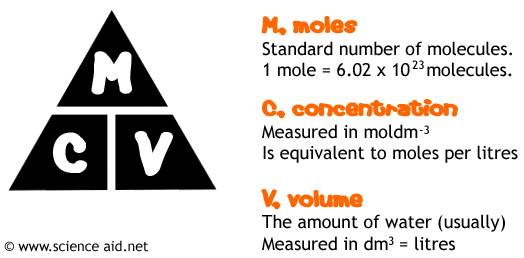
1.26 carry out mole calculations using volumes and molar concentrations
A solution contains a dissolved solute in a certain amount of solvent.
The concentration of a solution tells us how many moles of the solute are dissolved in one litre
(1 dm3) of the solvent.
(1 dm3) of the solvent.
The units for concentration are mol/dm3, and this is often shortened to M.
Concentration can also be measured in grams per litre.
n= v x c
Where v= volume (in dm3) and c= concentration (in mol/dm3)
NB: sometimes they give you the volume in cm3 so be careful, convert it to dm3 by dividing by 1000
(1 dm3 = 1000 cm3)
Or, you can just use: n= (v x c) / 1000
Remember to convert cm3 to dm3!
Solution
|
Formula
|
Concentration (mol m-3)
|
Concentration
(g dm-3)
|
Volume
|
Number of moles
|
Sodium hydroxide
|
NaOH
|
1
|
500 cm3
|
||
Hydrochloric acid
|
HCl
|
0.5
|
2 dm3
|
||
Sodium chloride
|
NaCl
|
58.5
|
4
|
||
Potassium chloride
|
KCl
|
200 cm3
|
0.2
|
||
Ammonium chloride
|
NH4Cl
|
0.25
|
250 cm3
|
||
Silver nitrate
|
AgNO3
|
2
|
0.5
|
||
Lithium iodide
|
LiI
|
||||
Sulphuric acid
|
H2SO4
|
0.2
|
1.8
|
||
Potassium nitrate
|
KNO3
|
150 cm3
|
0.15
|
- 25cm3 of a solution of 0.1M NaOH is neutralised by 50cm3 of HCl. What is the concentration of the HCl?
NaOH + HCl à NaCl + H2O
NB: remember to convert volumes to dm3!
n= v x c
n= 0.025 x 0.1=0.0025 mole
Mole ratio= 1:1, so moles of HCl= 0.0025 mole
C= n/v
C= 0.0025/ 0.05=0.05 mol/dm3
Practice:
- What mass of silver chloride precipitate will be produced if 25 cm3 of 0.1M silver nitrate is added to excess sodium chloride solution?(Answer may not be exact, so you give it to 2 significant figures here because the most exact data they give you here is to 2 sig. fig. which is the volume.)
- What mass of magnesium will react with 10cm3 of 1M HCl? Extra: and what volume of hydrogen will be formed? (1 mole of any gas at RTP is 24dm3)
Answers:
Substance
|
Formula
|
Mr
|
Mass of sample (g)
|
Number of moles in sample (n=m/Mr)
|
E.g. Water
|
H2O
|
18
|
9
|
0.5
|
Carbon dioxide
|
CO2
|
44
|
88
|
2
|
Ammonia
|
NH3
|
17
|
1.7
|
0.1
|
Sulphur dioxide
|
SO2
|
64
|
0.64
|
0.01
|
Sulphur trioxide
|
SO3
|
80
|
80
|
1
|
Hydrogen bromide
|
HBr
|
81
|
24.3
|
0.3
|
Sulphuric acid
|
H2SO4
|
98
|
0.098
|
0.001
|
Nitric acid
|
HNO3
|
63
|
3.15
|
0.05
|
Sodium nitrate
|
NaNO3
|
85
|
21.25
|
0.25
|
Sodium carbonate
|
Na2CO3
|
106
|
53
|
0.5
|
2 Na
|
+
|
2 H2O
|
à
|
2 NaOH
|
+
|
H2
|
46g
|
?
|
|||||
1:1 ratio
|
||||||
23g
|
:
|
40g
|
||||
46g
|
:
|
80g
|
||||
2. Calculate the mass of water formed when 32g of oxygen reacts with excess hydrogen.
2 H2
|
+
|
O2
|
à
|
2 H2O
|
1
|
:
|
2
|
||
32g
|
:
|
36g
(m = n x Mr
m = 2 x 18)
|
3. Calcium carbonate thermally decomposes to form calcium oxide and carbon dioxide. If 44g of carbon dioxide are collected, what mass of calcium oxide is formed?
CaCO3
|
à
|
CaO
|
+
|
CO2
|
1
|
:
|
1
|
||
56g
|
:
|
44g
|
||
*Remember that one mole of a substance is equivalent to its RFM in grams. For instance with CaO, the Mr of Ca is 40, and for O it is 16. Thus the RFM of CaO is 40 + 16 = 56. And since 44g of CO2 is 1 mole, then one mole of CaO must have been produced too (look at balanced equation), and 1 mole of CaO = 56g.
|
||||
Solution
|
Formula
|
Concentration (mol m-3)
|
Concentration
(g dm-3)
|
Volume
|
Number of moles
|
Sodium hydroxide
|
NaOH
|
1
|
40
|
500 cm3
|
0.5
|
Hydrochloric acid
|
HCl
|
0.5
|
18.25
|
2 dm3
|
1
|
Sodium chloride
|
NaCl
|
1
|
58.5
|
4 dm3
|
4
|
Potassium chloride
|
KCl
|
1
|
74.5
|
200 cm3
|
0.2
|
Ammonium chloride
|
NH4Cl
|
0.25
|
13.375
|
250 cm3
|
0.0625
|
Silver nitrate
|
AgNO3
|
2
|
340
|
0.25 dm3
|
0.5
|
Lithium iodide
|
LiI
|
1
|
134
|
1 dm3
|
1
|
Sulphuric acid
|
H2SO4
|
0.2
|
19.6
|
9 dm3
|
1.8
|
Potassium nitrate
|
KNO3
|
1
|
101
|
150 cm3
|
0.15
|
What mass of silver chloride precipitate will be produced if 25 cm3 of 0.1M silver nitrate is added to excess sodium chloride solution?(Answer may not be exact, so you give it to 2 significant figures here because the most exact data they give you here is to 2 sig. fig. which is the volume.)
AgNO3
|
+
|
NaCl
|
à
|
AgCl
|
+
|
NaNO3
|
1
|
:
|
1
|
||||
Moles of AgNO3
|
=
|
v
|
x
|
C
|
||
=
|
0.025
|
x
|
0.1
|
|||
=
|
0.0025 mol
|
|||||
Moles of AgCl
|
=
|
0.0025 mol
|
Due to 1:1 ratio
|
|||
m
|
=
|
n
|
x
|
RFM
|
||
=
|
0.0025
|
X
|
143.5
|
|||
≈
|
0.36g
|
(volume is only 2 sig. fig.)
|
||||
2. What mass of magnesium will react with 10cm3 of 1M HCl? Extra: and what volume of hydrogen will be formed? Assume RTP conditions -- 25°C and 1 atm (1 mole of any gas at RTP is 24dm3)
Mg
|
+
|
2 HCl
|
à
|
MgCl2
|
+
|
H2
|
1
|
:
|
2
|
:
|
1
|
||
Moles of HCl
|
=
|
v
|
x
|
C
|
||
=
|
0.01
|
x
|
1
|
|||
=
|
0.01 moles
|
|||||
Moles of Mg
|
=
|
0.005
|
(due to 1:2 ratio, 0.01 moles of HCl will react with 0.005 moles of Mg)
|
|||
Mass of Mg
|
=
|
n
|
x
|
RFM
|
||
=
|
0.005
|
x
|
24
|
|||
=
|
0.12g
|
|||||
1 mole of H2
|
=
|
24 dm3
|
=
|
24,000 cm3
|
||
0.005 mole of H2
|
=
|
0.005
|
x
|
24,000
|
||
=
|
120 cm3
|
|||||
So that's done.. hope the answers help. And good luck with your IGCSEs! I really can't update like this anymore, apologies but this was just to complete this post. Hope it helps anyway. :)
Are there answers anywhere?
ReplyDeletePlus , this blog is the best thing that happened to me during Christmas, Thanks very much
I don't think I actually put up answers but I can if you want them, do you still?
Deleteand hahahha, (y) nw, i'm glad :)
yess pleassssse
Deleteyou should put answers also....
Deletestill, this blog is very good source of information...
thank you for uploading this blog....
and please try to upload answers also............ :)
it is okay
DeleteTHIS IS THE BEST THING THAT HAS HAPPENED TO BE EVER, YOU DESERVE TO BE CROWNED, I LOVE YOU, OMG
ReplyDeleteyour nice comment is enough! :-)
DeleteThis is so helpful for revision for GCSE's iv emailed it to my entire class, thank you!
ReplyDeleteyou're welcome! and thanks for sharing my blog! :)
Deleteilysm i want to caress your sweet face you saved my life can i dedicate a blog to you can i give you my firstborn can u have both of my kidneys will u marry me
ReplyDeleteThis is very helpful. Thank you. It would help, if you had answers.
ReplyDeletethis blog is
ReplyDeletea m a z i n g.
thank you!
I was looking at the first answers table and
ReplyDeleteI'm not sure how the number of moles in the sample of sodium carbonate is 0.021?
I got m/Mr as 53/106.. so the answer was 0.5? :)
Ah yes you're right my bad! I forgot I changed some of the qs from my class exercise book to make the numbers easier, the mass in my book was 2.25g which gave 0.021..moles as an ans. (Didn't like the many decimal places so I changed the mass to 53, in which case yes the answer is 0.5 moles.)
DeleteSorry! And well spotted, will change it now. :D
Ok, just checking - thank you!! :) And thank you for this blog in general.. it is literally getting me through science revision at the moment!!! :D
DeleteYou're welcome :) Btw I thanked you at the bottom of the post haha (y) and good luck with your IGCSEs, I'm glad the blog helps! :D
Deletehow do you work out the mole ratio?! :s
ReplyDeleteYou look at the balanced chemical equation. For instance with:
DeleteMg + 2 HCl --> MgCl2 + H2
The ratio of Mg to HCl would be 1:2, since 1 mole of magnesium reacts with 2 moles of hydrochloric acid.
Does that make sense? :)
Wait, so why is it 1:1 in the example?
Deletebecause that mole ratio is of Mg to H2, you're trying to find the mass of hydrogen so that is what's relevant in this example qs. :)
DeleteOhhhhh! I get it! Thank you so much for answering! I finally get it :D
Deletei want these type of ques to come in my exam
ReplyDeletethey r soooooo easy
I love this site
its really very helpful
Can I say, though this is very helpful and your probably the best person for notes in IGCSE edexcel, what in the world makes you type so much and I think if thats a thing, I will be willing to pay a billion pounds for it.
ReplyDeleteHahhahha I shall take that as a compliment? Back during IGCSEs this was for my own revision + to help others, since I got so many questions this was the best way to help everyone. It did take ages, but it paid off. :) And now other years can use it :D
Deletethis sucks
ReplyDeletewho ever published this example can suck my cock..this is all bullshit i got to know from my teacher..SON OF A BITCH..WHO EVER DID THIS!!!!!!!
ReplyDeleteREPENT!!!
DeleteYOU SINNER, "I AM COMING SOON." WITH A REWARD FOR YOUR WORTHLESS TALK.
Thankyou for uploading this... this is one of the most useful mole calculation site on the web! Thanks!
ReplyDeleteI would like to recommend your article on Sodium Hydro sulphide. For further information, you can refer Sodium Hydro sulphide
ReplyDeleteIs the Mr for HBr2 81 or 161? Because on the first table in the answers section, that is what it says for the hydrogen bromide row.
ReplyDeleteMy apologies, it's meant to be 'HBr', I've changed it and the Mr would therefore be correct at 81. Thanks for spotting it. :)
DeleteSAVED MY LIFE
ReplyDeleteThis was a great help. :D Thank you sooo much!!!
ReplyDeletedont understand why '(due to 1:2 ratio, 0.01 moles of HCl will react with 0.005 moles of Mg)' works in the 2nd volumes question
ReplyDeletecan u put some more question for practice
ReplyDeleteThis was very useful! thank you :)
ReplyDeleteamazing blog
ReplyDeletewhy in Mg+2HCl in the end we are dividing 0.01 by 2 other then multiplying????????
ReplyDeletethank you, it really helps... I like the way everything is explained
ReplyDelete0.005*24 does not equal 0.012g in the magnesium question. Correct mass should be 0.12
ReplyDeleteThanks so much!!! It has helped me a lot....hope I get good marks in my exam tomorrow.. Thanks!
ReplyDeleteSorry to bother you but I was just wondering for the first answers table, the answer for Sodium Bromide is different to what I got and I was really confused.
ReplyDeleteSo Moles = Mass/Mr so therefore Moles = 24.3/81 which is = 0.3 but for your answer it was 0.22 so I was just wondering if I did something wrong...? Thank you for this Blog, it saved my life
Hi there, if you are referring to the Hydrogen Bromide answer then you are quite right! It's my bad. I'll change it, thanks for spotting that! And no problem, really glad the blog helps. :)
Deletecan u plssolve this 4 me
ReplyDeleteThank You so much!!!! I never got this topic until i looked on this blog!! Its incredibly helpful!!!
ReplyDeleteHi, the last question 2 just before the comments has the answer of 0.012 however it is actually 0.12. Just thought I should let you know :)
ReplyDeleteThank you!! :)
DeleteWELCOME :)
Deleteguys
ReplyDeletechill this topic is easy
YEET! ITS A GUD BLOG FAM!
ReplyDeleteMy Chemistry exams are tomorrow, I’ve been trying to understand moles coz everything went bouncer, your blog helped me a lot, hoping I pass. Good luck for your blog, pretty sure I’ll check your blog when I have doubts. Gracias
ReplyDeleteYou saved me sir as moles was a dreadful chapter like a bad dream for me but because of you I can score nice in moles in my mock examination
ReplyDeleteon topic 7 question number 2 where are you getting the other number of males
ReplyDeletehi thank you for this but I have a question for the first practice question in the answer part
ReplyDeleteHi!
ReplyDeleteThank you so much for these! I'm preparing for IGCSEs and I was stuck on this topic.